|
Niels Bohr proposed an early model of the atom as a central nucleus containing protons and neutrons. In Bohr’s model of the hydrogen atom, one electron, carrying a negative electrical charge, circles a nucleus consisting of a single proton, which has a positive. The quantum mechanical model specifies the probability of finding an electron in the three-dimensional space around the nucleus and is based on solutions of the Schrödinger equation. Bohr Diagrams of Atoms and Ions Electron Shells. The Bohr model assumes that the electrons move in circular orbits that have quantized energies, angular momentum, and radii that are specified by a single quantum number, n 1, 2, 3,, but this quantization is an ad hoc assumption made by Bohr to incorporate quantization into an essentially classical mechanics description of the atom. Ultimately, models based entirely on quantum mechanics replaced the Bohr model. 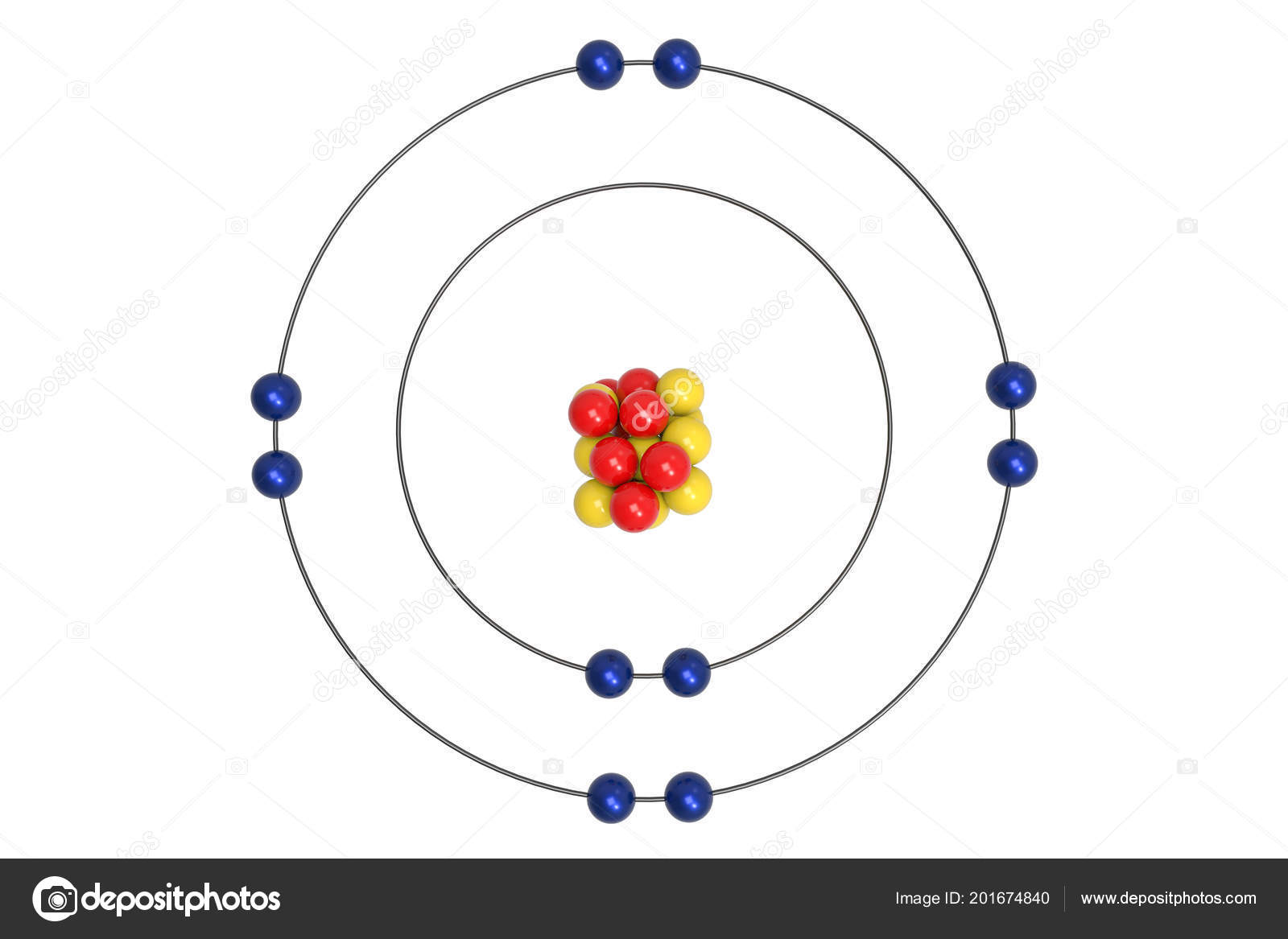
Earlier models were the cubic model (1902), plum-pudding model (1904), Saturnian model (1904), and Rutherford model (1911). The quantity in parentheses is a unit of energy known as a Rydberg, which \(\approx 13.6eV\).īohr reasoned that the hydrogen atom doesn't radiate away all of its energy, because the lowest energy level (which we would now call the ground state) corresponds to one wavelength fitting in the orbit, so \(n=1\) is the lowest it can go. An atomic orbital, which is distinct from an orbit, is a general region in an atom within which an electron is most probable to reside. The Bohr model was the first atomic model incorporating some quantum mechanics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
